T cells can be redirected towards pre-defined target cells by engineering with a chimeric antigen receptor (CAR) for use in adoptive cell therapy of malignant diseases. We here used the strategy to explore and manipulate the functional interaction of engineered antigen-redirected T cells with solid tumors and tissue stem cells.
The adoptive cell therapy of malignant diseases has shown powerful in the treatment of hematologic malignancies in early phase trials. T cells of patients are genetically engineered with a chimeric antigen receptor (CAR) which confers pre-defined specificity by an antibody and T cell activation by signaling through domains of the T cell receptor complex. We used CAR T cells to address a number of basic issues on T cell – tumor and stem cell interaction.
Complete eradication of large established solid tumors has not been reported to date. Although CAR-Ts recognize the antigen MHC-independently and exclusively on cancer cells without targeting the cross-presented antigen, we revealed complete eradication of large established HER-2+ tumors by HER-2 specific CAR-Ts. Tumor rejection occurred without the recognition of the cross-presented antigen but only when CAR-Ts were provided with the CD28 costimulatory signal, however, it required the expression of IFNγ receptor and Fas on the tumor stroma.
Textor et al., Cancer Res (2014)
Given the tremendous phenotypic diversity in solid tumor lesions, a reasonable number of cancer cells are not recognized by a given CAR. We described a strategy for overcoming this shortcoming of the CAR T cell therapy by modulating the tumor stroma by a CAR T cell secreted transgenic cytokine like IL-12. The basic process is that CAR T cells, when activated by their CAR, deposit IL-12 in the targeted tumor lesion, which in turn attracts an innate immune cell response towards those cancer cells that are invisible to CAR T cells. Such TRUCKs, T cells redirected for universal cytokine-mediated killing, exhibited remarkable efficacy against solid tumors with diverse cancer cell phenotypes suggesting their evaluation in clinical trials.
Chmielewski et al., Immunol Rev (2014)
Chmielewski et al., Exp Opin (2015)
One bottleneck for adoptive T cell therapy is recruitment of T cells into tumors. We showed that combining tumor-specific T cells, modified with a marker antigen and a bispecific antibody (BiAb) that selectively recognizes CAR T cells and tumor cells improves T cell recruitment to tumors and enhances therapeutic efficacy with increased T cell infiltration of tumors, retarded tumor growth, and prolonged survival.
Kobold et al., J. Natl. Cancer Inst. (2015)
In solid tumors T cells encounter a hostile environment, in particular with increased inflammatory activity that goes along with abundant reactive oxygen species (ROS) that substantially impair antitumor activity. We present a strategy to render CAR T cells more resilient toward ROS by coexpressing catalase to increase their anti-oxidative capacity by metabolizing H2O2. In fact, T cells engineered with catalase and CAR performed superior over CAR T cells as they showed increased levels of intracellular catalase and had a reduced oxidative state with less ROS accumulation while maintaining their antitumor activity despite high H2O2 levels. Moreover, CAR-CAT T cells exerted a substantial bystander protection of other immune effector cells. Bystander NK cells, otherwise ROS sensitive, efficiently eliminate their target cells under oxidative stress when admixed with CAR-CAT T cells. This approach represents a novel means for protecting tumor-infiltrating cells from tumor-associated oxidative stress-mediated repression.
Ligtenberg et al., J Immunol (2016)
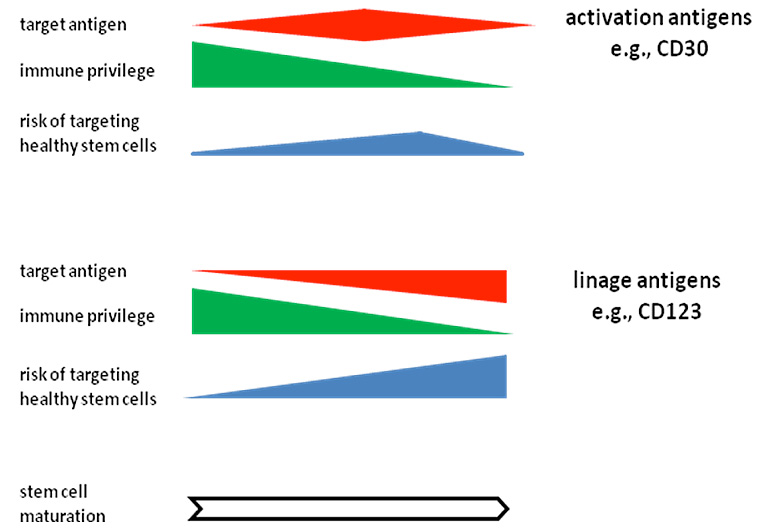
Targeting B lymphoma/leukemia by anti-CD19 CAR T cells is effective, however, eliminates healthy B cells. We here explored CD30 as an alternative target, which is validated in lymphoma therapy and expressed by a broad variety of lymphomas. Targeting CD30 also targets hematopoietic stem and progenitor cells (HSPCs). We revealed that HRS3scFv-derived CAR T cells were not blocked by soluble CD30 and did not attack CD30+ HSPCs while eliminating CD30+ lymphoma cells. Consequently, normal hemato- and lymphopoiesis was not affected in the long-term in the humanized mouse; the number of blood B and T cells remained unchanged. We provide evidence that the CD30+ HSPCs are protected against a CAR T-cell attack by substantially lower CD30 levels than lymphoma cells and higher levels of the granzyme B inactivating SP6/PI9 serine protease, which furthermore increased upon activation.
Hombach et al., Mol Ther (2016); Expert Rev Clin Immunol (2016)
Adoptive cell therapy of chronic lymphocytic leukemia (CLL) with CAR T cells targeting CD19 induces lasting remission in a number of patients, however, is associated with prolonged “on-target off-tumor” toxicities due to the targeted elimination of healthy B cells. We identified the immunoglobulin M Fc receptor (FcmR) as a target for a more selective treatment of CLL. CAR T cells specific for FcmR efficiently responded toward CLL cells, released proinflammatory cytokines, eliminated the leukemic cells and delayed outgrowth of Mec-1-induced leukemia in a xenograft mouse model. CAR T cells from CLL patients in various stages of the disease purged their autologous CLL cells in vitro without reducing the number of healthy B cells implying a superior therapeutic index of anti-FcmR CAR T cells for the treatment of CLL.
Faitschuk et al., Blood (2016)
Chmielewski et al., (2014) Immunol. Rev. 257, 83 - 90
Faitschuk et al., (2016) Blood 128, 1711 – 1722
Faitschuk et al., (2016) Gene Therapy, in press
Hombach et al., (2016) Mol. Ther., 24, 1423 – 1434
Kobold et al., (2015) J. Natl. Cancer Inst. 107, 364 - 371
Krug et al. (2015) Cancer Immunol. Immunother. 64, 1623 – 1635
Krug et al., (2014) Cancer Immunol. Immunother. 63, 999-1008
Ligtenberg et al., (2016) J. Immunol. 196, 759-766
Makalowski et al., (2014). In: Cancer Stem Cells. Chapter 33, Ed.: V. K. Rajasekhar, Wiley-Blackwell, p. 451 – 460
Textor et al., (2014) Cancer Res. 74, 6796 – 6805
Information from this funding period will not be updated anymore. New research related information is available here.

CMMC Affiliation
associated RG (05/2018-06/2019) Principal Investigator B 01 (1/2017-05/2018)
show more…+49 941 943 5980
+49 941 943 5932
CMMC Affiliation
present address: Am Biopark 9
93053 Regensburg
http://www.rcii.de/forschung/lehrstuehle/lehrstuhl-fuer-gen-immuntherapie/
Curriculum Vitae (CV)
Markus Chmielewski (Post Doc)
Astrid Holzinger (Post Doc)
Andreas Hombach (Post Doc)
Johannes Kühle (Post Doc)
Dorottya Horváth (PhD student)
Valérie Liebrecht (PhD student)
Elena Faitschuk (PhD student)
Jennifer Makalowski (PhD student)
Alexandra Martyniszyn (PhD student)
Viktória Golumba-Nagy (marie-curie, PhD student)
Lisa Hannappel (PhD student)
Anja Meyer (Master student)
Carola Janke (Bachelor student)
Danuta Chrobok (Technician)
Nicole Hoffmann (Technician)
Petra Hofmann (Technician)
Birgit Hops (Technician)