
Successful scientific teamwork: Anne Wolf (PhD student and first author) | from left to right: Dr. Michael Schramm | Univ.-Prof. Dr. Thomas Langmann | Dr. Marc Herb | Image: Christoph Wanko - Uniklinik Köln
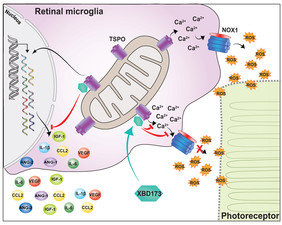
Figure legend: In response to retinal damage, microglia increase pro-inflammatory and pro-angiogenic cytokine expression and upregulate mitochondrial TSPO leading to increased cytosolic calcium levels, which is essential for NOX1-mediated ROS production that is toxic for photoreceptors. Binding of the synthetic ligand XBD173 to TSPO limits inflammatory responses and inhibits the increase of cytosolic calcium levels thus preventing from ROS damage. XBD173 also supports the conversion of reactive microglia towards a neuroprotective phenotype, thereby limiting pathological angiogenesis in the eye.
In the study the researchers report that a protein not previously associated with AMD, but known in the brain as a biomarker for inflammation, the so-called translocator protein (TSPO), was identified as a signal molecule for the release of oxygen radicals in the damaged retina.
The work was published in the renowned scientific journal Nature Communications. Wolf A. Herb M, Schramm M, Langmann T.: The TSPO-NOX1 axis controls phagocyte-triggered pathological angiogenesis in the eye. Nature Communications 2020; DOI: 10.1038/s41467-020-16400-8
In a large number of degenerative retinal diseases that lead to blindness, chronic immune activation takes place in the eye. Microglia cells are particularly aggressive in age-related macular degeneration (AMD). AMD a multifactorial retinal degenerative disease leads to severe vision loss amongst the elderly. The number of AMD patients is expected to reach 196 million worldwide by 2020 and increase to 288 million by 2040. Early AMD is characterized by drusen deposits in the subretinal region of the macula. Late-stage AMD can manifest either as geographic atrophy (GA) (dry form) or as the wet form characterized by choroidal neovascularization (CNV). While the etiology of AMD is still not well understood, genome-wide association studies and experimental animal models have unequivocally shown dysregulated innate immune responses in AMD involving complement and mononuclear phagocytes, including local microglia.
In the healthy retina, microglia constitute a surveillance network in the plexiform layers to preserve tissue integrity. After injury, they proliferate and migrate to the site of damage to phagocytose cell debris, releasing neuromodulatory factors to promote tissue repair. While a short period of controlled activation is considered to be neuroprotective, chronic microglia reactivity may endanger the already compromised retinal tissue.
Therefore, the release of proinflammatory cytokines and reactive oxygen species (ROS) and the concomitant phagocytosis of healthy neurons can be a driving force for photoreceptor demise and disease progression.
The translocator protein (18 kDa) (TSPO) is described as a biomarker for reactive gliosis, but its biological functions in retinal diseases remain elusive. In this study, the team focused on elucidating the molecular function of TSPO in retinal immune homeostasis and angiogenesis.
“Rather casually, I told Dr. Marc Herb and Dr. Michael Schramm from the Institute of Microbiology, Immunology and Hygiene about our project”, Anne Wolf, the PhD student of Langmann´s team and first author of the publication: “and this was the starting point of our cooperation."
“Michael Schramm and Marc Herb are both experts in the field of immune mediators and it became clear very quickly, that oxidative stress and reactive oxygen species were the major harmful products of microglia cells”, Thomas Langmann comments, who is heading the Laboratory of the Immunology of the Eye strongly affiliated with the Center for Molecular Medicine Cologne (CMMC).
In the laser-induced mouse model of neovascular AMD the researchers could show that tamoxifen-induced conditional deletion of TSPO in resident microglia using Cx3cr1CreERT2:TSPOfl/fl mice or targeting the protein with the synthetic ligand XBD173 prevents reactivity of phagocytes. Concomitantly, the subsequent neoangiogenesis and vascular leakage are prevented by TSPO knockout or XBD173 treatment. Using different NADPH oxidase-deficient mice, the researchers were able to demonstrate that TSPO is a key regulator of NOX1-dependent neurotoxic ROS production in the retina. These findings of the first preclinical immunotherapy experiments with TSPO inhibitors in animal models are considered as proof of principle showing the protection against retinal degeneration and pathological vascularization in the eye. The data show also that neurotoxic ROS production in microglia is regulated by a TSPO-mediated increase in calcium levels and activation of NADPH oxidase 1 (NOX1).
“The identification of the important and distinct role for TSPO in ROS production of phagocytes provides a molecular mechanism for TSPO/ROS-related immunomodulatory and neuroprotective therapies in the retina and the brain. We hope that our findings will accelerate the development of new treatment options for elderly patients with vision loss”, Thomas Langmann comments.
Original pulication:
Wolf A. Herb M, Schramm M, Langmann T.: The TSPO-NOX1 axis controls phagocyte-triggered pathological angiogenesis in the eye.
Nature Communications 2020; DOI: 10.1038/s41467-020-16400-8 | https://www.nature.com/articles/s41467-020-16400-8
Scientific Contact:
Prof. Dr. Thomas Langmann
Laboratory for Experimental Immunology of the Eye Department of Ophthalmology and
Center for Molecular Medicine Cologne
Faculty of Medicine and University Hospital Cologne, University of Cologne, Cologne, Germany.
thomas.langmann[at]uk-koeln.de
http:/expimmeye.uni-koeln.de
https://www.cmmc-uni-koeln.de/research/research-areas-projects/research-area-b/thomas-langmann-khalid-rashid-b-05
CMMC News Release:
Dr. Debora Grosskopf-Kroiher
Center for Molecular Medicine Cologne
debora.grosskopf-kroiher[at]uni-koeln.de