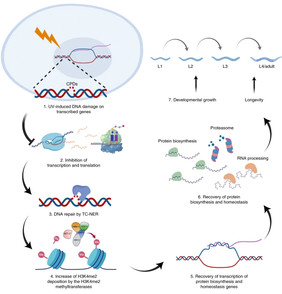
Model of the role of H3K4me2 in the regulation of UV-induced DDR. Transcription-blocking DNA damage leads to the inhibition of transcription and translation. After the completion of NER, H3K4me2 is deposited by the WRAD-regulated MLL/COMPASS complex and SET-17, followed by the recovery of transcription of protein-biosynthesis and homeostasis-related genes. Through increasing protein biosynthesis and re-establishing homeostasis, the animals resume development and longevity post-DNA damage. This figure was created with BioRender (https://biorender.com/).
The research team led by Björn Schumacher, heading the Institute for Genome Stability in Aging and Diseases and Principal Investigator of the Cluster of Excellence for Aging Research CECAD and the Center for Molecular Medicine Cologne (CMMC) at the University of Cologne (Faculty of Medicine) has discovered that a change in the DNA structure – more precisely in the chromatin – plays a decisive role in the recovery phase after DNA damage. The key is a double occupation by two methyl groups on the DNA packaging protein histone H3 (H3K4me2). The specific change enables genes to be reactivated and proteins to be produced after damage: The cells regain their balance and the organism recovers. The protective role of H3K4me2 was identified in experiments with the nematode Caenorhabditis elegans.
The study uncovers a role for H3K4me2 in coordinating the recovery of protein biosynthesis and homeostasis required for developmental growth and longevity after DNA damage and has now been published in the journal Nature Structural & Molecular Biology:
The genome in every human cell is damaged on a daily basis, for example in the skin by UV radiation from the sun. DNA damage causes cancer, impairs development and accelerates aging. Congenital malfunctions in DNA repair can lead to extremely accelerated aging in rare hereditary diseases. Therefore, preservation and reconstruction processes are particularly important to ensure development and to maintain tissue function. DNA, which is rolled up on packaging proteins – the histones – like on cable drums, is regulated by methyl groups. Various proteins are responsible for placing methyl groups on histones or removing them. The number of groups on the packaging proteins affects the activity of genes and thus the protein production of the cell.
In experiments with the nematode, the researchers showed that after repairing damaged DNA, two methyl groups were increasingly found on the DNA packages. Furthermore, they found that errors in placing these two methyl groups on the histones (H3K4me2) accelerated the damage-induced aging process, while increased position of this histone alteration prolongs the lifespan after DNA damage. By controlling the proteins that either set or remove these methyl groups, the resistance to DNA damage – and thus the aging process of the animals – could be influenced. Further analysis of the role of these two methyl groups showed that the enrichment of H3K4 after genome damage with two methyl groups supports the cells in restoring the balance after DNA damage.
The reseachers report that, in Caenorhabditis elegans, removal of the WRAD complex of the MLL/COMPASS H3K4 methyltransferase exacerbates developmental growth retardation and accelerates aging, while depletion of the H3K4 demethylases SPR-5 and AMX-1 promotes developmental growth and extends lifespan amid ultraviolet-induced damage.
The researchers demonstrate that DNA-damage-induced H3K4me2 is associated with the activation of genes regulating RNA transport, splicing, ribosome biogenesis and protein homeostasis and regulates the recovery of protein biosynthesis that ensures survival following genotoxic stress.
"Now that we know the exact changes in chromatin, we can use this to precisely limit the consequences of DNA damage,’ Schumacher comments and adds: "I hope that these findings will enable us to develop therapies for hereditary diseases characterized by developmental disorders and premature aging. Due to the fundamental importance of DNA damage in the aging process, such approaches could also counteract normal aging and prevent age-related diseases."
Publication
S. Wang, D. Meyer & B. Schumacher. H3K4me2 regulates the recovery of protein biosynthesis and homeostasis following DNA damage. (2020) Nature Structural & Molecular Biology.
doi: 10.1038/s41594-020-00513-1 / https://www.nature.com/articles/s41594-020-00513-1
Contact
Prof. Dr. Björn Schumacher
Institute for Genome Stability in Aging and Diseases - Faculty of Medicine, University of Cologne
bjoern.schumacher[at]uni-koeln.de
CMMC news release = modified press release of University of Colognefrom Dr. Anna Euteneuer
https://portal.uni-koeln.de/en/universitaet/aktuell/press-releases/single-news/forschungsteam-entdeckt-mechanismus-der-die-zellfunktion-nach-genomschaeden-wiederherstellt